
Everyone deserves to live the best life that they can, as free as possible from the challenges and uncertainty of disease.
Driven by that vision, UCB’s journey in rare and ultra rare diseases began with a deeply personal moment: a UCB scientist, frustrated by the limited treatment options available for his wife’s rare autoimmune condition, used his expertise to help transform her care.
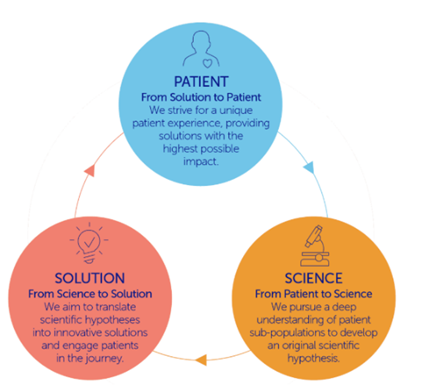 Decades later, that same ethos of “Inspired by patients, Driven by science” has resulted in an achievement few can claim: three FDA-approved therapies in just three years, each addressing areas of significant high unmet need.
Decades later, that same ethos of “Inspired by patients, Driven by science” has resulted in an achievement few can claim: three FDA-approved therapies in just three years, each addressing areas of significant high unmet need.
Our first two approvals in targeted treatments in generalized myasthenia gravis (gMG) marked critical steps toward expanding the the UCB rare disease portfolio. Today, we are proud to announce that KYGEVVI™, the first and only FDA-approved treatment for thymidine kinase 2 deficiency (TK2d) in adult and pediatric patients with an age of onset on or before 12 years, is now commercially available by prescription in the United States.
UCB is committed to patient access and is offering ONWARD®, our patient support program, to help simplify the process of getting eligible patients started on their prescribed therapy of KYGEVVI.
With ONWARD, each patient will be assessed at the individual level to understand their coverage.The patient will be paired with a dedicated Care Coordinator, who will help explain potential financial assistance options that could help.* †
Three approvals in three years is more than just a milestone. It reflects what is possible when we put the rare and ultra-rare disease patient at the heart of everything that we do.
From Patient to Science
Alexis had a terrible sinus infection that unexpectedly progressed to double vision, paralysis in one eye, and eventually a diagnosis of generalized myasthenia gravis. For him, everyday activities as simple as walking, brushing his teeth, and cooking for his family became daily challenges.
Levee was just four months old when he rapidly lost physical abilities and experienced full respiratory failure before he was diagnosed with TK2d.
By listening deeply to patient journeys like these, we gain a clearer understanding of their lived realities — insights that help us design solutions that close the care gap and address the unmet needs of these sub-populations in rare disease communities.
From Science to Solution
Scientific innovation matters only when it reaches the people who need it. That’s why UCB remains committed to expanding access and delivering meaningful impact beyond regulatory milestones.
Closely aligning our goals with the needs of the rare disease community, UCB’s approach to innovation and partnership informs not just our treatments, but also our wider contributions to support and care.
Every new approval and launch strengthens our commitment to create value for the rare and ultra-rare disease patient communities that we serve, now and into the future. They deserve care, no matter how rare.
* ONWARD is provided as a service of UCB and is intended to support the appropriate use of UCB medicines. ONWARD may be amended or canceled at any time without notice. Some program and eligibility restrictions may apply.
† ONWARD does not provide medical advice and does not replace the care of the healthcare provider. Care Coordinators will refer you to your healthcare provider for any treatment-related questions.
This content is intended for a U.S. audience only.
About KYGEVVI
KYGEVVI is a combination of doxecitine and doxribtimine, both pyrimidine nucleosides, indicated for the treatment of thymidine kinase 2 deficiency (TK2d) in adults and pediatric patients with an age of symptom onset on or before 12 years.1 Administration of KYGEVVI is intended to incorporate the pyrimidine nucleosides, deoxycytidine and deoxythymidine, into skeletal muscle mitochondrial DNA.1 This action restores mitochondrial DNA copy number in TK2d mutant mice.1
Important Safety Information for KYGEVVI1
Increase in Liver Transaminases
Elevated liver transaminase [alanine aminotransferase (ALT) and/or aspartate aminotransferase (AST)] levels were reported in patients treated with KYGEVVI. Obtain baseline liver transaminase (ALT, AST) and total bilirubin levels in patients prior to treatment initiation with KYGEVVI. If signs or symptoms consistent with liver injury are observed, interrupt treatment with KYGEVVI until liver transaminase (ALT, AST) and total bilirubin levels have either returned to baseline or stabilized at a new baseline value. Consider permanently discontinuing KYGEVVI if signs or symptoms consistent with liver injury persist or worsen. Monitor liver transaminases and total bilirubin levels yearly and as clinically indicated.
Gastrointestinal Adverse Reactions
Diarrhea and vomiting leading to hospitalization, dose reduction, and permanent discontinuation were reported in patients treated with KYGEVVI. Based on the severity of the diarrhea and/or vomiting, reduce the dosage of KYGEVVI or interrupt treatment until diarrhea and/or vomiting improves or returns to baseline. Consider restarting KYGEVVI at the last tolerated dose, and increase the dose as tolerated. For persistent or recurring diarrhea and/or vomiting, consider discontinuing KYGEVVI permanently and provide supportive care with electrolyte repletion as clinically indicated.
Adverse Reactions
The most common adverse reactions (incidence ≥5%) are diarrhea, abdominal pain (including abdominal pain upper), vomiting, alanine aminotransferase increased (ALT), and aspartate aminotransferase increased (AST).
Please see the full U.S Prescribing Information for additional information. Talk to your healthcare provider about your condition or your treatment.
Reference:
- KYGEVVI [Prescribing Information]. Smyrna, GA: UCB, Inc.
KYGEVVITM and ONWARD® are (registered) trademarks of the UCB Group of Companies.
©2026 UCB, Inc., Smyrna, GA 30080. All rights reserved. US-MT-2600001