
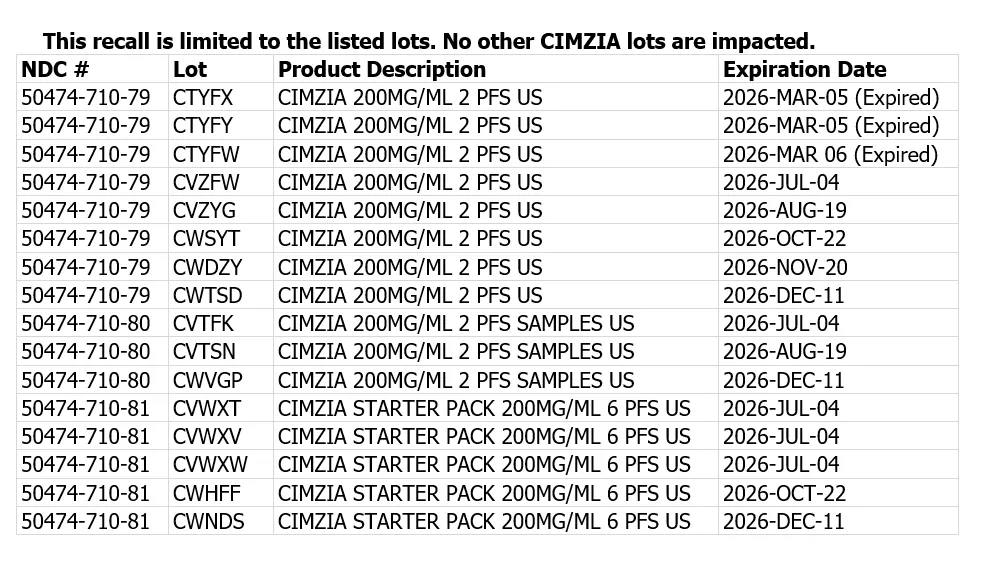
UCB has initiated a voluntary recall in the United States of 16 lots of CIMZIA® (certolizumab pegol) pre-filled syringes (PFS). UCB received reports of broken or cracked syringes, which may break during handling or use, for select CIMZIA PFS product distributed from November 2024 through February 2026.
Reported incidents related to damaged syringes have been limited, and UCB continues to monitor and encourage reporting of any adverse events or quality problems. Based on UCB’s investigation, the observed issue relates to the syringe and does not affect the safety or effectiveness of the medicine contained within it.
As part of our commitment to high quality standards and patient safety, UCB initiated this voluntary recall in close coordination with the U.S. Food and Drug Administration (FDA) and our distribution partners to support continuity of care. UCB has also identified the root cause of the issue and has taken steps to prevent future lots from being impacted.
There is no anticipated impact on CIMZIA supply.
Products Affected
- Select lots of CIMZIA® (certolizumab pegol) pre-filled syringes (PFS)
- U.S. market only
- Distributed from November 2024 through February 2026
The following CIMZIA products are NOT affected by this recall:
- CIMZIA lyophilized (LYO) administrated via in-office injection
- CIMZIA Polyarticular Juvenile Idiopathic Arthritis (JIA)
The lot number can be found on the CIMZIA carton (Figure 1) and device (Figure 2):

Products from the affected lots should not be used and should be returned. UCB is notifying our distributors and customers and is arranging for return of all recalled products. Distributors and healthcare providers should review inventory for affected lots and follow recall instructions as provided to return to the point of purchase for replacement.
Questions about the recall should be directed to UCBCares® by phone +1-844-599-CARE (2273) or email UCBCares@ucb.com.
Patients should speak with their healthcare provider about individual care and before adjusting their CIMZIA treatment.
Adverse reactions or quality problems experienced with the use of this product may be reported to UCBCares or to the FDA's MedWatch Adverse Event Reporting program either online, by regular mail or by fax.
- Complete and submit the report online: www.fda.gov/medwatch/report.htm
- Regular mail or fax: Download form www.fda.gov/MedWatch/getforms.htm or call
1-800-332-1088 to request a reporting form, then complete and return to the address on the pre-addressed form, or submit by fax to 1-800-FDA-0178
Full Prescribing Information, including boxed warning regarding serious infections, including tuberculosis, and certain malignancies.
Contact Information
For questions or additional information, please contact:
UCBCares®
UCBCares@ucb.com
+1-844-599-CARE (2273)
www.ucb-usa.com/Patients
For media or IR inquiries, please contact:
Nicole Herga
U.S. Communications
T +1 773.960.5349
nicole.herga@ucb.com
Yvonne Naughton
Head of Investor Relations
T +44.175.344.7521
yvonne.naughton@ucb.com
Sahar Yazdian
Investor Relations Lead
T +32.2.559.91.37
sahar.yazdian@ucb.com
Choose a Country
- Global Site – English
- Australia – English
- België – Engels
- Belgique – Anglais
- Brasil – Português
- България – Български
- Canada – English
- Canada – Français
- 中国 – 中文
- Česká Republika – Angličtina
- Danmark – Engelsk
- Deutschland – Deutsch
- France – Français
- España – Español
- Ελλάδα – Ελληνικά
- India – English
- Ireland – English
- Italia – Inglese
- 日本 – 日本語
- Казахстан – ағылшын тілі
- 한국 – 한국어
- Luxembourg – Anglais
- Luxemburg – Engels
- Magyarország – Angol
- México & Latinoamérica – Español
- Nederland – Engels
- New Zeeland – English
- Norge – Engelsk
- Österreich – Deutsch
- Polska – Polski
- Portugal – Inglês
- România – Engleză
- Россия – Русский
- Slovensko – Anglický
- Suomi – Englanti
- Sverige – Engelska
- Schweiz – Deutsch
- Suisse – Français
- Türkiye – Türkçe
- Україна – Англійська
- United Kingdom – English
- U.S.A. – English